 The Y group acts as a leaving group during the nucleophilic acyl substitution reaction.The acyl group is the remainder of the molecule, which includes the carbonyl and the attached alkyl group.Carboxylic acid derivatives contain an electronegative heteroatom (typically oxygen, nitrogen, sulfur, or phosphorus) bonded directly to the carbonyl carbon represented by the symbol Y.Acyl phosphates are named following the IUPAC rules with the carboxylic acid ending with -oyl or -yl and -phosphate.Ģ1.2 Nucleophilic Acyl Substitution Reactions.Cyclic amides (lactams) use a Greek letter to identify the location of the nitrogen in relation the carbonyl group. 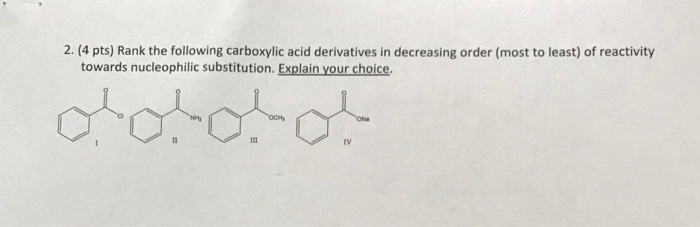
Secondary or tertiary amides are named as primary amides are, but in additon an upper case N is used to designate the alkyl groups attached to the nitrogen atom, which are names as substituents.Primary amides are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -amide. The reactivity order is as follows: Acid chloride > anhydride > thioester > ester > amide Most reactive least reactive There is a very large range of reactivity between the most reactive (acid chlorides) and the least reactive (amides).Thioesters are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -thioate and the sulfide alkyl chain as a substituent.Esters are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -ate and the alkoxy alkyl chain as a substituent.Anhydrides are named following the IUPAC rules with the carboxylic acid ending -acid being replaced by -anhydride.Acid halide are named following the IUPAC rules with the carboxylic acid ending with -oyl or -yl and the halide ending in an -ide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
